creation of hydrazine

Theodor Curtius, a German chemist, was the first person who came into contact with the hydrazine molecule. Theodor Curtius spent his early years in Germany studying the subject of chemistry at the University of Heidelberg. Much later in the year 1889, Curtius became the Director of Analytical Chemistry at the University of Erlangen. Soon after, Mr. Theodor Curtius made a baffling discovery. N2H4, also referred to as hydrazine, was synthesized in a lab and formed with the help of the Olin Raschig Process. The Raschig process reacts pure ammonia (NH3) with hypochlorite (ClO), and the two together form the highly toxic substance known as hydrazine. It was soon discovered that the substance created is highly flammable and must be handled with serious caution only under strict surveillance. Along with these risks came the dangerous consequences, and hydrazine poisoning was introduced. More recently, hydrazine is used as liquid propellant fuel for rockets, but is starting to prove unsanitary for the environment. Although the liquid is capable of being stored for lengthy periods of time, it is becoming too big of a risk for both workers and the environment to handle. A research study has been sent out by NASA Corporation in search for a new, more eco-friendly alternative to the ever-so dangerous hydrazine liquid propellant rocket fuel. An astounding amount of fifty million dollars is currently being offered up to anyone who is able to create an alternative rocket fuel for the NASA Corporation.
What is hydrazine?
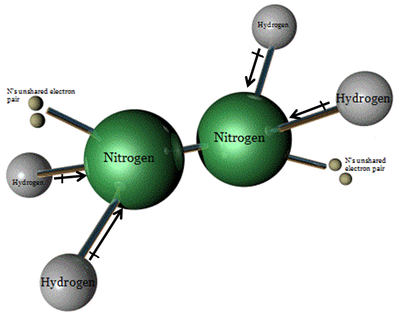
Hydrazine has a chemical formula of N2H4 and is formed from a reaction between ammonia and hypochlorite. It was initially used as a reducing agent in polymer foams, yet the current use is far more fascinating. Hydrazine is used as a component in rocket fuel, and in the past has been used for airplane fuel as well. Hydrazine was and is used most commonly by NASA because of its efficient characteristics. Hydrazine is an oily, transparent liquid that is said to have a bit of an ammonia-like odor. Depending on concentration levels, the odor is capable of becoming more pungent. From the years 1988 to 2003, over 500,000 pounds of hydrazine was disposed of, mainly by the means of underground injection wells. The rest of this excess hydrazine was naturally oxidized into the environment and biodegraded if at a low concentration. Over the past 100 years, times have changed and the uses of hydrazine have differed greatly. A few sections down is a brief timeline of hydrazine applications in history.
Specifics
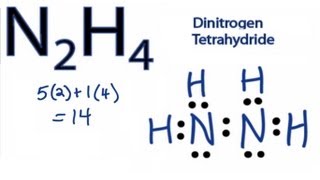
Formula: N2H4
Molar Mass: 32.0452 g/mol
Melting Point: 35.6 degrees Fahrenheit
Boiling Point: 237.2 degrees Fahrenheit
Density: 1.02 g/cm^3
Molar Mass: 32.0452 g/mol
Melting Point: 35.6 degrees Fahrenheit
Boiling Point: 237.2 degrees Fahrenheit
Density: 1.02 g/cm^3
Hydrazine structural formula
hydrazine applications timeline (1930-1976)
Handling, storage, and current usage
Hydrazine is currently used at NASA as a liquid propellant for rockets. It must be handled under strict surveillance and only by qualified professionals. Specific suits must be worn when it is being handled, as it is highly corrosive. If hydrazine comes in contact with skin, severe burning may result and long-term damage will be enforced. NASA has released a public statement in the past year stating that the company is searching for a new, more beneficial and "green" rocket fuel as hydrazine is quite a dangerous substance for handling. It is, however, capable of being stored for long periods of time without being seriously affected. Hydrazine is not overall good to be releasing into the environment, and a new fuel will soon be in the works. Below are three pictures of hydrazine handling by NASA professionals.
Hydrazine is also used as a corrosion inhibitor in boiler water systems. In this way, hydrazine slows down the corrosion rate of a certain material, causing it to be in use for a longer period of time. Hydrazine is a highly flammable substance, and should be stored in well-labeled, tight containers. It should never come into contact with direct sunlight, and the storage container should be ventilated and contain a working drainage system that runs underground in case of spills or leakage. Hydrazine should be stored at no higher or lower than 250° C, as that is when it is in its most stable and calm state.
Hydrazine is also used as a corrosion inhibitor in boiler water systems. In this way, hydrazine slows down the corrosion rate of a certain material, causing it to be in use for a longer period of time. Hydrazine is a highly flammable substance, and should be stored in well-labeled, tight containers. It should never come into contact with direct sunlight, and the storage container should be ventilated and contain a working drainage system that runs underground in case of spills or leakage. Hydrazine should be stored at no higher or lower than 250° C, as that is when it is in its most stable and calm state.

In the mid-1900s, a derivative of hydrazine posed as a reliable and consistent treatment for Tuberculosis and is still in use today. Isoniazid, a mixture of hydrazine and isonicotinic acid with a chemical formula of C6H7N30, is a medication given either by injection or orally to treat Tuberculosis infections. There are possible long-term and short-term side effects to the medication, very similar to those of hydrazine poisoning. Some side effects include nausea, vomiting, sore throat, fever, itching/swelling, all of which can eventually but rarely lead to liver disease typically in adults of age 35 or older. Isoniazid is overall the second leading treatment for the Tuberculosis virus, and there is both a four month treatment and two month treatment option.
dangers and toxicity of hydrazine
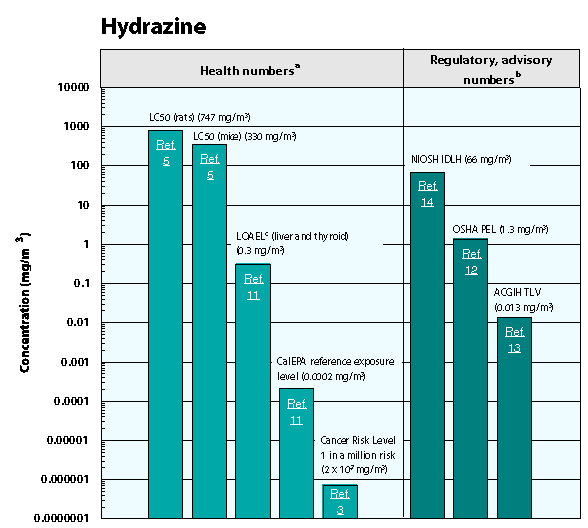
Hydrazine has a reputation for being highly corrosive, and is a known carcinogen. It is also highly flammable and is very dangerous when exposed to the average everyday human, both orally and externally. Hydrazine poisoning is most commonly treated with the deliverance of vitamin B and 10% dextrose under medical care. However, when mixed with certain other ingredients in a lab setting, hydrazine is capable of being orally ingested and used as medication in the form of Isoniazid, as mentioned above.
Below is a timeline of what one could expect if faced with hydrazine poisoning:
Immediately after oral ingestion:
Below is a timeline of what one could expect if faced with hydrazine poisoning:
Immediately after oral ingestion:
- Burning eyes
- Burning skin
- Vomiting
- Slurring of words
- Unconsciousness
- Inability to breathe correctly
- Severe violence spells
- Problems with pupils within eyes
- Convulsing
- Comatose state