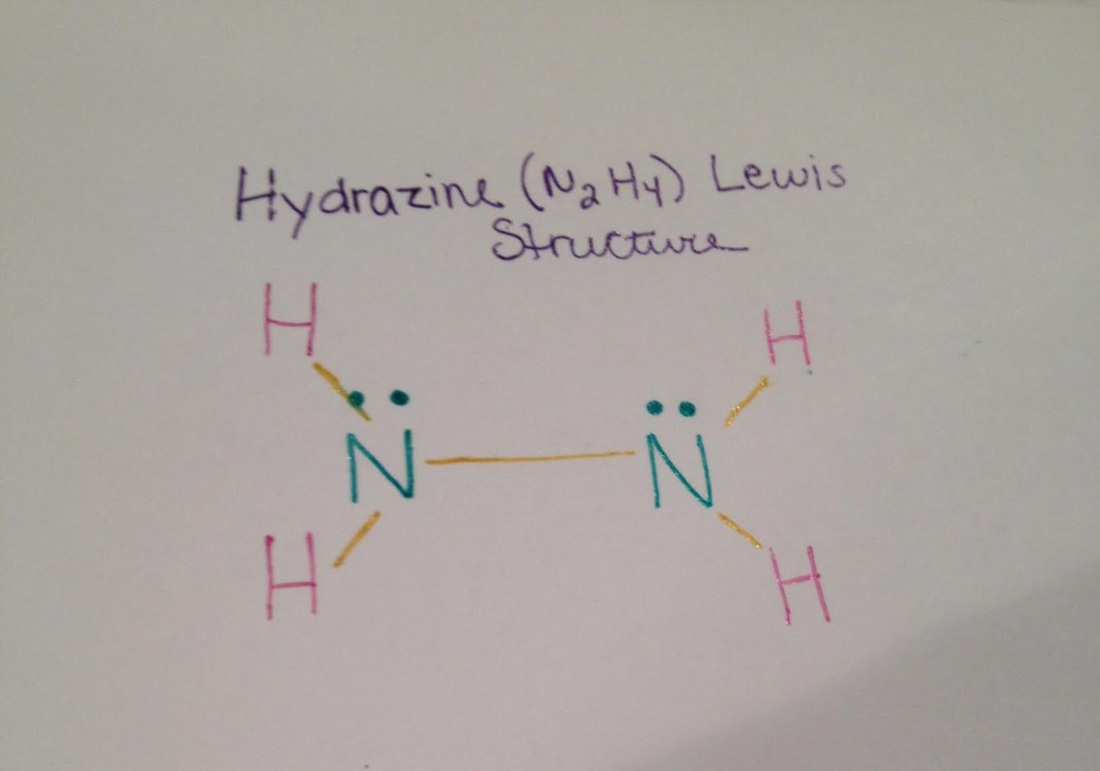
Hydrazine lewis structure
The Hydrazine Lewis Structure is combined of two Nitrogen atoms, each attached to each other and two Hydrogen atoms. In total, hydrazine is comprised of 6 atoms, two of which (Nitrogens) each have their own pair of unshared electrons.
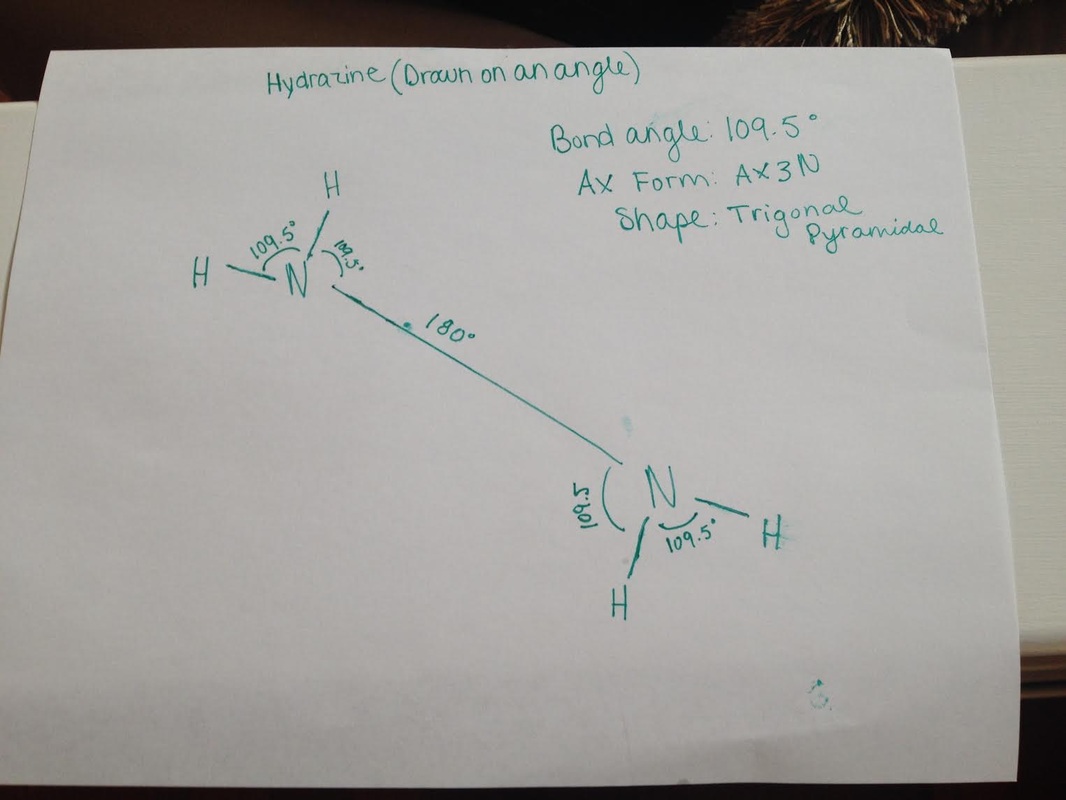
hydrazine drawing and polarity
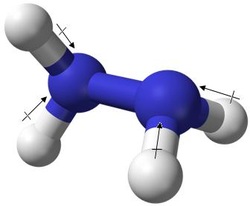
Hydrazine is classified as a polar molecule, the reason being that polarity from the lower energy level (Hydrogen) is being distributed into that of the higher energy level (Nitrogen), which therefore creates an uneven distribution of energy and charges. No bonds are capable of being created between the two nitrogen, as the bond is fully covalent. The two nitrogen atoms shown in the drawing each have a pair of unshared (lone) electrons. These lone electrons cause the nitrogen atoms to take on a slightly negative charge, while the four hydrogen have positive charges.