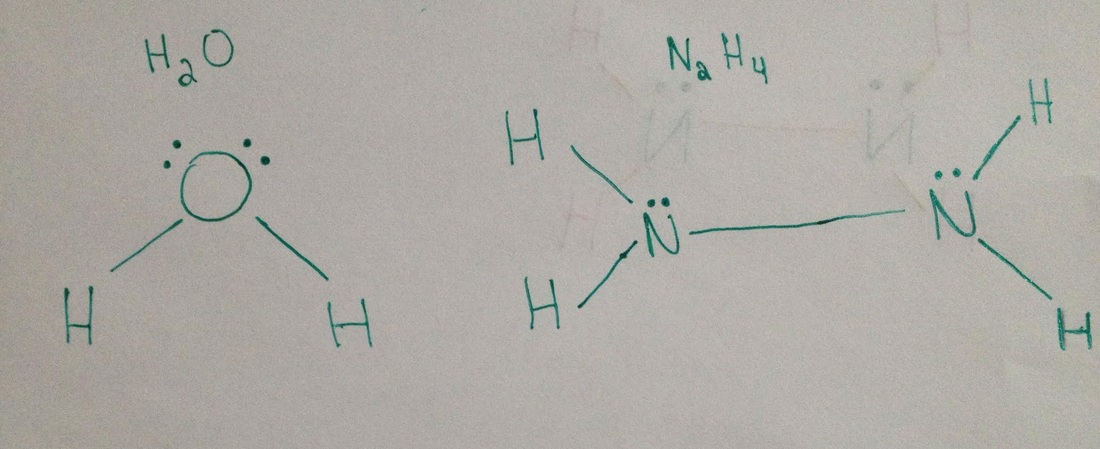
Intermolecular forces between n2h4 and h2o
A water molecule (H2O) has been placed next to a hydrazine molecule (N2H4) to show an example of the intermolecular forces that are capable of acting upon a single hydrazine molecule. There are several intermolecular forces acting between the hydrazine and water molecules.
- Between these two molecules, it can be said that there is dipole-dipole interaction present as an intermolecular force. Both the water and hydrazine molecules are polar, therefore there is dipole-dipole interaction. The positive hydrogens of the N2H4 molecule are attracted the the negative oxygen molecule of the H2O molecule. Dipole-dipole interaction only occurs between two polar molecules, but only under certain circumstances. The two molecules cannot be perfectly symmetrical, or non-polar, for this intermolecular force to be present, but rather be asymmetrical, or polar.
- There are also London (Dispersion) Forces acting upon these two molecules. London forces occur between and two adjacent molecules when the two liquids are in close contact with one another. These forces are present around the surrounding areas of the two molecules, and are scattered freely across their entirety.
- There is also Hydrogen bonding between the two molecules shown above. Hydrogen bonding requires an H atom to be initially bonded to either Nitrogen, Oxygen, or Fluorine and from there it is attracted to the N, O, or F of another molecule. In the above situation, the hydrogen atom from N2H4, which is bonded to nitrogen, interacts with the oxygen from the H2O molecule, creating the final intermolecular force known as Hydrogen Bonding.